News
News
Featured Products
| REF | 502010 | Specification | 20 Tests/Box |
| Detection principle | Immunochromatographic assay | Specimens | Whole Blood/Serum / Plasma |
| Intended Use | StrongStep® H. pylori Antibody Rapid Test is a rapid visual immunoassay for the qualitative presumptive detection of specific IgM and IgG antibodies to Helicobacter pylori with human whole blood/serum/plasma as specimen. | ||
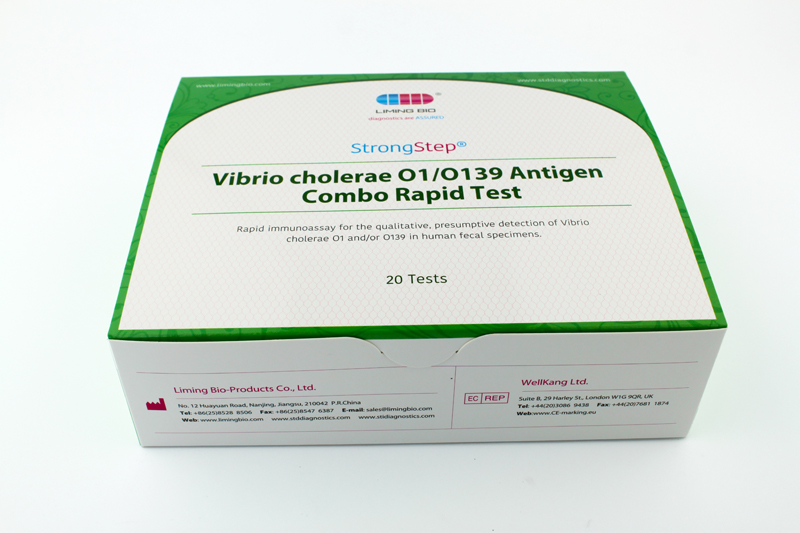
| REF | 501070 | Specification | 20 Tests/Box |
| Detection principle | Immunochromatographic assay | Specimens | Feces |
| Intended Use | The StrongStep® Vibrio cholerae O1/O139 Antigen Combo Rapid Test is a rapid visual immunoassay for the qualitative, presumptive detection of Vibrio cholerae O1 and/or O139 in human fecal specimens. This kit is intended for use as an aid in the diagnosis of Vibrio cholerae O1 and/or O139 infection. | ||
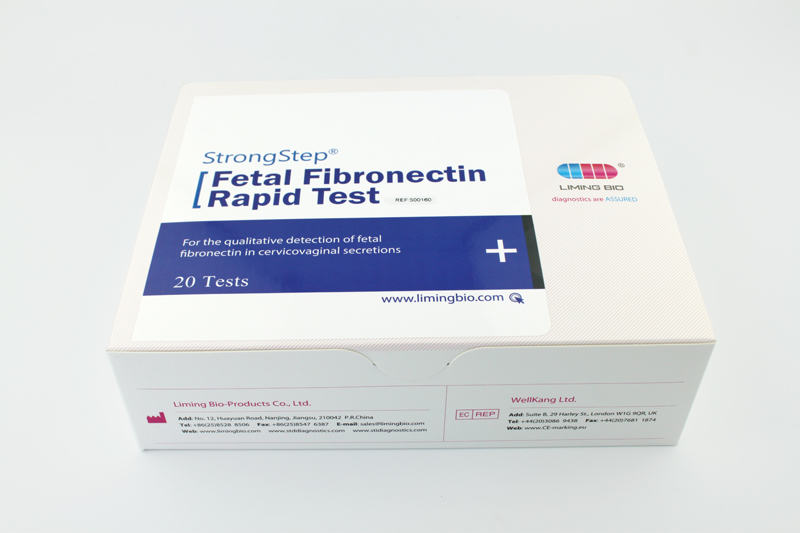
| REF | 500160 | Specification | 20 Tests/Box |
| Detection principle | Immunochromatographic assay | Specimens | Cervicovaginal secretions |
| Intended Use | StrongStep® Fetal Fibronectin Rapid Test is a visually interpreted immunochromatographic test intended to be used for the qualitative detection of fetal fibronectin in cervicovaginal secretions. | ||
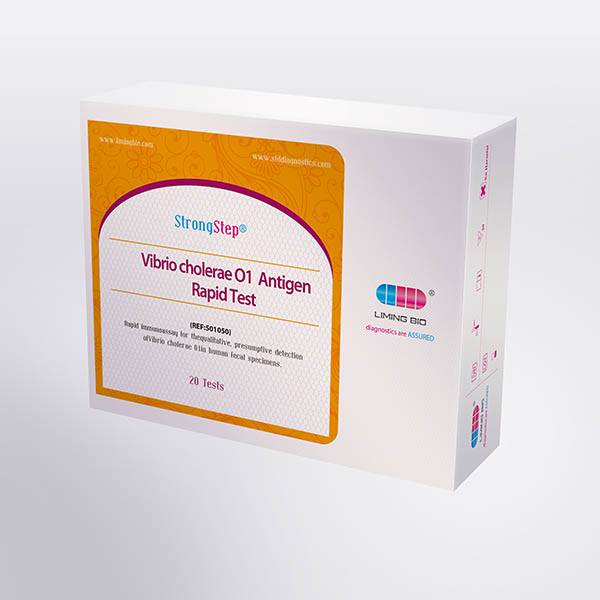
| REF | 501050 | Specification | 20 Tests/Box |
| Detection principle | Immunochromatographic assay | Specimens | Feces |
| Intended Use | The StrongStep® Vibrio cholerae O1 Antigen Rapid Test Device (Feces) is a rapid visual immunoassay for the qualitative, presumptive detection of Vibrio cholerae O1 in human fecal specimens. This kit is intended for use as an aid in the diagnosis of Vibrio cholerae O1 infection. | ||
| REF | 500170 | Specification | 20 Tests/Box |
| Detection principle | Immunochromatographic assay | Specimens | Vaginal discharge |
| Intended Use | StrongStep® PROM rapid test is a visually interpreted, qualitative immunochromatographic test for detection of IGFBP-1 from amniotic fluid in vaginal secretions during pregnancy. | ||
| REF | 500020 | Specification | 20 Tests/Box |
| Detection principle | Immunochromatographic assay | Specimens | Cervical/urethra swab |
| Intended Use | It is suitable for qualitative detection of gonorrhoea/chlamydia trachomatis antigens in cervical secretions of women and urethral samples of men in vitro in various medical institutions for auxiliary diagnosis of the above pathogen infection. | ||